CBSE Class 6 Science – Curiosity – Takshila-DPS Pune- Patna-Coimbatore
AUTHOR
MBBS, DNB (DVL)
Dermatologist, Cosmetologist and Laser Surgeon
Nuleaf Skin Clinic and Hair Transplant Center, Undri, Pune
As a mother of twin daughters and a Consultant Dermatologist based in Undri, Pune, life is always a balancing act. Between busy clinic hours, parenting duties, and everyday chaos, I’ve been creating study notes since Class 4 to help my girls understand and revise their lessons better. Over the years, these weekend study sessions turned into a special tradition—discussing topics taught in school and turning them into organized, easy-to-revise notes.
The notes I’m sharing here are from the 2024–25 academic year of DPS Pune – Patna – Coimbatore, and cover Class 6 chapters. They include all the key points that are likely to be asked in exams, compiled from the Class 6 Curiosity textbook, school study materials, class notes and school revision sheets.
Each page was made with care, love, and a hope to make learning feel a little easier—especially during exam time. If they help your child too, then this effort finds even more meaning.
If you find these notes useful, I’d love to hear from you in the comments section. And if you’d like me to upload notes on any particular topic from Class 6, feel free to send in your suggestions.
Don’t forget to subscribe to my blog for new updates, notes, and posts.Wishing you and your child a wonderful academic year ahead!
Warmly,
Gauri
IMPORTANT POINTS
- 7,000 to 8,000 years ago: The earliest pottery found in the Indian subcontinent in the Ganga plains (Lahuradewa) and in Baluchistan (Mehrgarh).
- About 4000 BCE onwards: Sindhu-Sarasvatī developed techniques of wheel turned pottery production, pigmentation, application of protective or decorative coats (called ‘slips’) of multiple colours, decorative painting, etc.
- 2600–1900 BCE:These techniques became further sophisticated during the Sindhu-Sarasvatī (also known as ‘Harappan’) Civilisation with a bright red surface painted with black-coloured designs displaying geometric patterns, and aquatic and terrestrial animals.
- The clay used for making pots, dishes, bowls and other items was carefully selected and cleaned, sieved, kneaded, turned over a wheel and finally baked in kilns (baked clay is called ‘terracotta’).
- Pots were used for various purposes, from cooking to storage of food grains, oil, ghee, and so on.
- Some very large storage jars and other pottery items are exhibited at the National Museum, New Delhi.
NOTES
Grouping things
Q) Why do we need to group things?
Ans) Grouping of things or materials is very helpful in our day-to-day life.
It helps to reduce the chances of things getting mixed up and it becomes very easy to locate a particular thing.
Example:
- A chemist arranges various medicines either alphabetically or by company name. If he keeps these medicines randomly (without any grouping) it would be very difficult to locate the required medicine.
- In a book shop, books are arranged on the basis of class & the subjects.
- In a grocer’s shop, you’ll find that the eatables are kept apart from soaps and shampoos
Classification
Classification (Defn): The process of arranging of things into groups on the basis of their common properties is called classification.
Advantages of Classification:
- The classification of objects into groups makes it easier to locate them and work with them.
- Classification of objects makes it convenient to study properties of object of one category and also observe any pattern in these properties.
(This is because if we know the properties of any one member of the group, we can get an idea of the properties of the other members of this group.)
Basis of classification of objects
The classification of various objects into groups can be done on the basis of their similarities and dissimilarities such as:
- Living or non-living
- Plant or animal
- Nature of material
- Use
- Colour, Shape, Size;
- Appearance
- Hardness
- Texture etc.
Material:
Material (Defn): A substance (type of matter) which is used to make different objects is called material.
Example: a chair is made of wood, so wood is a material.
We choose a material to make an object depending on:
- its properties and
- the purpose for which the object is to be used.
Materials can be used in the following ways:
- A material can be used to make different kinds of objects/ things.
Example: glass can be used to make tumblers, bowls, mirrors, window-panes etc.
- An object may be made up of a combination of materials.
Example: a table can be made up of wood, metal, glass.
- Same thing can be made from different kinds of materials,
Example: bowls can be made from stainless steel, glass, plastic, thermocol & porcelain.
- Materials may be naturally-occurring or man-made.
Example: Wood, cotton, jute, marble etc. are naturally occurring materials. Glass, steel, thermocol, plastic, polyester, nylon etc. are man-made materials.
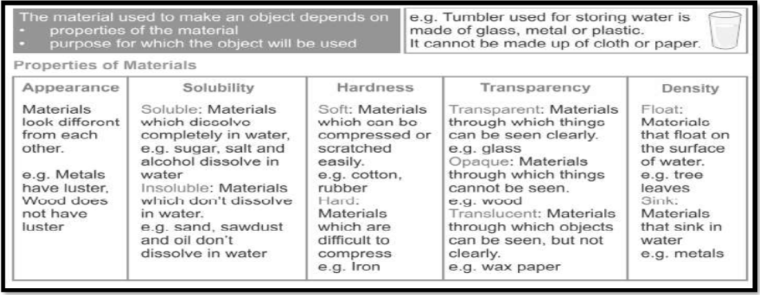
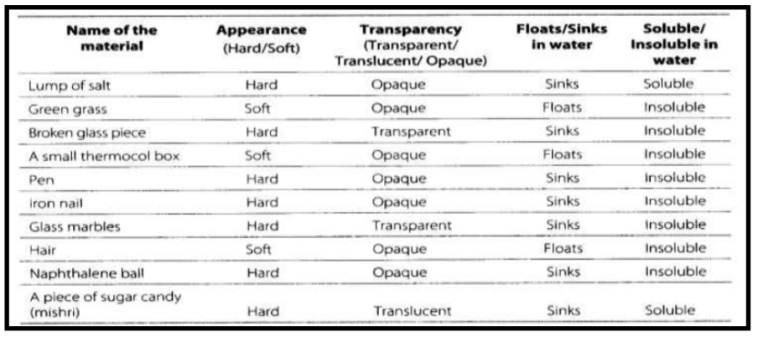
Properties of materials:
Property (Defn): A property is the description of a substance that helps to identify it.
1. LUSTRE (SHINE)
(Defn): Materials that shine are called lustrous, and those which do not are called non-lustrous.
Examples:
- All metallic substances, e.g. materials used for making jewellery, utensils, cutlery, water taps, door handles, flower vases etc. are lustrous.
- Iron, copper, zinc, silver, aluminium and gold are examples of metals.
Characteristics:
- The characteristic shine of metals is called metallic lustre.
- On exposure to humid air, most metals lose their lustre in the course of time, and then the metal is said to be tarnished.
- The metal regains its lustre if its surface is rubbed with cloth or sandpapers or polished.
- Surfaces of some materials are made shiny by polishing or coating them with thin layers of plastic, wax or any other material which makes them look shiny. These materials may not be metals.
Q) Why do we notice lustre only on freshly cut surface of metals?
Ans) Some metals often lose their shine and appear dull, because of the action of air and moisture on them. We therefore, notice the lustre, only on their freshly cut surface.
2. HARDNESS
(Defn): Materials which are difficult to compress i.e. whose shape cannot be changed by pressing are called hard whereas materials which can be compressed or scratched easily and whose shape can be easily changed by pressing or pulling apart are called soft.
Examples: A hard material is used to make a table, chair or bed while a soft material must be used to make a cushion or clothing.
Characteristics:
- Diamond is the hardest natural substance known. It can cut glass.
- Cotton or sponge is soft because it traps lot of air in it.
- Iron is hard
- These properties are relative in nature. For example, rubber is harder than sponge but softer than iron.
| Hard material | Soft material | ||
| 1 | It cannot be compressed | 1 | It can be compressed easily |
| 2 | It is difficult to scratch or tear | 2 | It is easy to scratch or tear |
| 3 | It can be broken if force is applied | 3 | It gets squeezed when force is applied |
| 4 | Example: Chalk, stone, iron | 4 | Example: Wool, sponge |
3. SMOOTHNESS
(Defn): Materials with an even surface are smooth while those whose surface is uneven are rough.
Examples: The surface of the mirror or stainless-steel bowl is smooth whereas that of the stone or brick is rough.
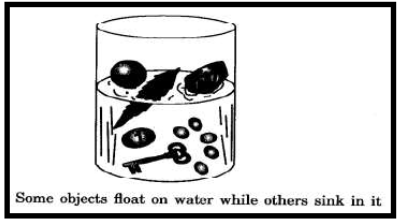
4. FLOATING OR SINKING IN WATER
A material lighter than water floats, whereas those which are heavier than water sinks, for example, paper and plastic float, whereas sand and iron sink.
5. CONDUCTIVITY
(Defn): Materials can conduct electricity or act as insulators.
Example: Metals like copper and aluminium are good conductors of electricity, while rubber and plastic are insulators
6. SOLUBILITY IN WATER
Definitions:
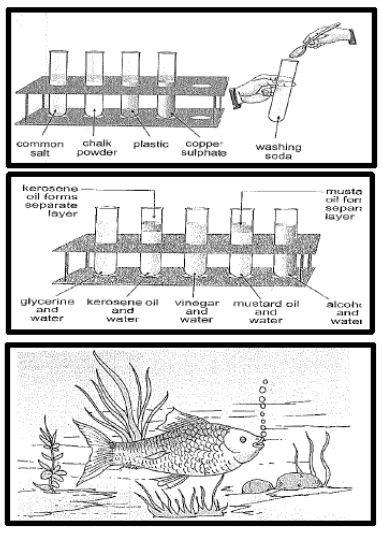
- Soluble/Insoluble: The materials that dissolve in water are said to be soluble and the materials that do not dissolve even after stirring for a long time are said to be insoluble.
- Solute/Solvent: A liquid that dissolves other substances is called a solvent and the substance that is dissolved is called a solute.
For most of solutes the solubility increases with increase in temperature.
- Solution: The homogeneous mixture containing the solute and solvent is called a solution.
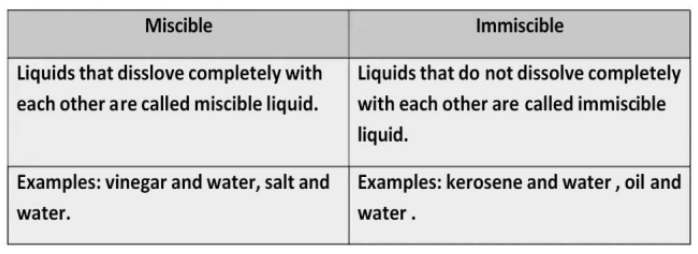
- Miscible/ Immiscible:
- Solubility: The maximum amount of solute in grams which can be dissolved in 100 grams of the solvent at a given temperature to form a saturated solution is called the solubility of the substance. It depends on the physical and chemical properties of the solute and solvent as well as on temperature.
Examples:
- Some liquids, like glycerin and shampoo, mix with water to form a solution. Such liquids are said to be miscible with water.
Liquids like edible oil or kerosene, which do not mix with water, are said to be immiscible with water. Immiscible liquids, when mixed, form separate layers.
- Some gases like oxygen and carbon dioxide are also soluble in water. Aquatic plants and animals use the carbon dioxide and the oxygen dissolved in water for their survival.
- Aerated water (soft drinks) contains dissolved carbon dioxide. Gases like nitrogen, methane and helium do not dissolve in water.
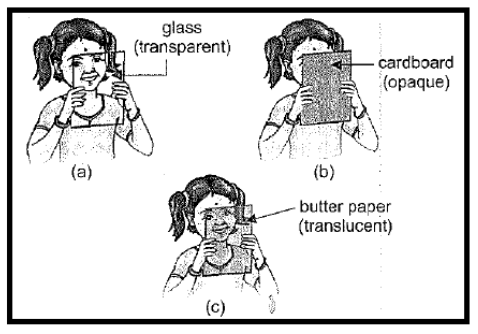
8. TRANSPARENCY
Different materials behave differently when light falls on them.
| Transparent materials | Opaque materials | Translucent materials | |
| 1 | The materials which allow light to pass through them are called transparent materials. | The materials which do not allow light to pass through them are called opaque materials. | Certain materials allow only a part of the light to pass through them and are called translucent materials. |
| 2 | Through transparent materials objects can be seen clearly. | Through Opaque materials objects cannot be seen at all. | Through translucent materials objects can be seen, but not clearly |
| 3 | Glass, cellophane paper, acrylic sheet, water, air, some plastics etc. | Wood, stone, butter paper, frosted glass, cardboard, metals etc. | Oily paper or waxed paper, ground glass, frosted glass etc. |
9. DENSITY
(Defn): Density is defined as mass per volume.
- You can think of it as the numbers of particles of a substance are packed into a certain amount of space.
- If the particles are packed tightly together, the density would be greater than if they are loosely packed with a lot of empty space around them.
- This is taken as mass per unit volume of a given object.
- The SI unit is kg/m3.
- Density is the reason some objects sink, and other objects float.
10. MATTER
(Defn): Anything that occupies space and has mass is called matter.
11. MASS
Mass (Defn): Mass is the quantity of matter contained in an object or a body.
- Mass and volume are the two properties that are possessed by all materials.
- Mass considered as one of the fundamental quantity since all the objects in the universe contain mass.
- We can say that any object which is heavier or lighter can be measured in terms of a property called mass.
- The one which is heavier has more mass and the one which is lighter has less mass.
- Mass quantifies the amount of matter present in an object.
Weight (Defn): Weight is the gravitational force experienced by the mass of an object.
Units for mass
- The SI unit of mass is kilogram (kg).
- Usually, for measuring mass, the units of a kilogram are preferred over the gram units.
- Heavyweights are measured in tonne or metric tonne.
- 1 gram (g) =1000 milligram (mg)
- 1 kilogram (kg) =1000 gram (g)
- 1 tonne =1000 kilogram (kg)
Difference between Mass and Weight:
| Mass | Weight | ||
| 1 | Mass is the quantity of matter contained in an object or a body. | 1 | Weight is the gravitational force experienced by the mass of an object. |
| 2 | The mass of an object is a constant value and does not vary by any change in gravity or place. | 2 | The value of weight changes depending on thegravitational pull or location. |
| 3 | The mass may be measured using an ordinary balance. | 3 | Weight is measured using a spring balance |
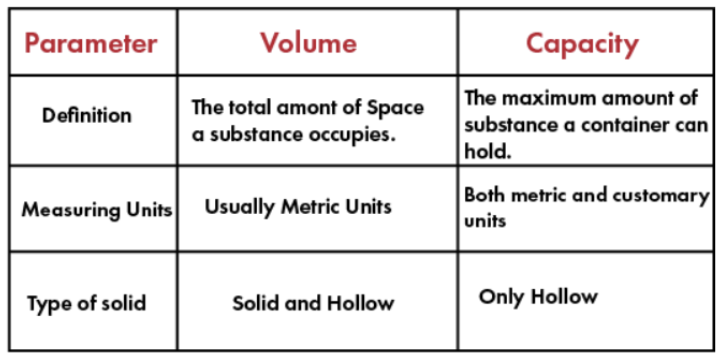
12. VOLUME AND CAPACITY
Volume (Defn): The volume is the measure of space the matter occupies.
(OR) The space occupied by matter is its volume.
Capacity (Defn): The capacity of a container is defined as the maximum amount of substance it can hold.
For example, if you pour 2 litres of water in a 5 litre bottle, then the volume of water in the bottle is 2 litres and the capacity or volume of the bottle is 5 litres.
Units: Volume and capacity are measured in metric units such liters and milliliters as well as customary units such as gallons, pints, cups and quarts.
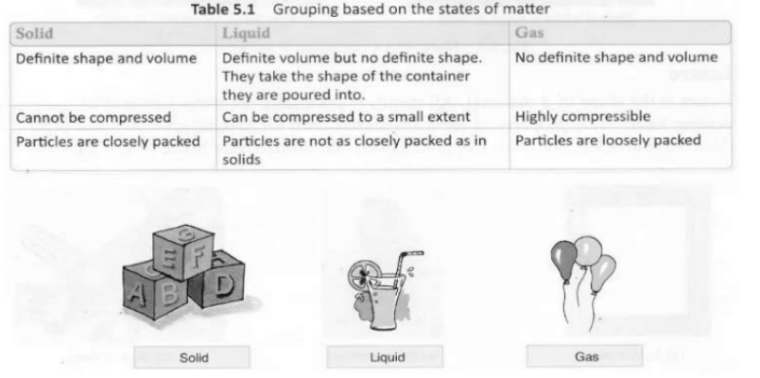
States of matter
All substances are made up of matter. On the basis of physical state, materials or matter exist in three states: solid, liquid and gas.
Examples:
(i) Solid – sand, steel and stone.
(ii) Liquid -oil, water and milk.
(iii) Gas – steam, nitrogen and oxygen.
Properties of materials
- Material occupy space
- Materials have mass
To read:
Kilogram is the unit of mass in the International System of Units (SI). Kilogram is abbreviated in lower case as kg. There is no space between ‘k’ and ‘g’ in kg, and no full. stop after the symbol, except at the end of a sentence. While writing the mass, always leave a space between the number (numerical value) and the unit. For example, if we have mass of 7 kilograms, it would be written as 7 kg and not as 7 kgs. Similarly, litre is abbreviated as capital L and millilitre as mL. There is no space between ‘m’ and ‘L’ in mL. For example, if you have 500 millilitres of water, it would be written as 500 mL, m will be in lower case and L will be in upper case. The SI unit forvolume is cubic metre, abbreviated as m3. The abbreviation is written with a superscript 3 to denote cubic metre. For example, if you have volume of 2 cubic metres, it would be writtenas 2 m3. Always leave a space between number(numerical value) and the unit. 1 m3 =1000 L.
Thanks a lot for visiting my blog. Please do ‘★ LIKE’ the post if you found it to be useful. Your comments, suggestions, criticism and all opinions are very much appreciated. Please do write your queries in the Reply/comments section and I will try to get back to you asap.

All the content on this blog is copyrighted. It takes immense research, hard work, patience and time to come up with a comprehensive article/post. If you like and need to use any of my article(s) or any part of it, please do give the requisite reference, link or credit to my website/blog . DO NOT COPY WITHOUT PERMISSION.